Combustible Nano-Dust: Smaller Particle Sizes Lead to Faster, Stronger Explosions
By: Timothy Cullina, Senior Consulting Engineer, Fauske & Associates
 Credit for launching the nanotechnology revolution frequently goes to Richard Feynman’s 1959 talk to the American Physical Society, “There’s Plenty of Room at the Bottom”. And even if you disagree with this credit, I hope you can agree that Feynman’s enthusiasm for “small science” certainly fed the imaginations of scientists, philosophers, and deep thinkers about a future built upon nanotechnologies.
Credit for launching the nanotechnology revolution frequently goes to Richard Feynman’s 1959 talk to the American Physical Society, “There’s Plenty of Room at the Bottom”. And even if you disagree with this credit, I hope you can agree that Feynman’s enthusiasm for “small science” certainly fed the imaginations of scientists, philosophers, and deep thinkers about a future built upon nanotechnologies.
Others, maybe Eric Drexler, or was it Omni Magazine, forecast a darker future for nanotechnology with predictions of self-replicating nanobots gone rogue, consuming nearly all resources, and leaving only a dark, dead, world composed of nothing but useless nanobots forming endless piles of “grey goo”.
More than a half century later, the nano engines of creation are quietly humming away, the alarms over grey goo have gone nearly silent, and many of today’s industries move steadily toward producing smaller particulate, even if most are still quite a ways from true nanometer particle size.
Particle size is the dominant physical parameter that affects explosion severity and ease of ignition for combustible dusts. And particulate size is trending down in many industries. This is true for pigment, toners, electronics, cosmetics, pharmaceuticals, specialty chemicals, additive manufacturing, food and many more. As with toner, the reason may be improved quality and new transfer technologies. Other advantages may be related to improving mixing efficiencies and reducing production times.
Of course, some particle size reductions may go unnoticed if your management of change (MOC) program is blind to particle size considerations. While there are advantages to working with a smaller particle size that is specific to each application, there is also an increased combustible dust risk associated with smaller particle sizes that needs to be managed.
For example, 10 years ago printer toner particulate sizes averaged 30 to 50 microns. To improve the appearance of type, manufacturers have worked to produce finer and finer toner particulate. Today, toner manufacturers are pushing the size boundaries to, and even below, 10 microns. Ten years ago, KSt values for many toners were in the Class II range, that is 200 to 300 bar-meter per second. Today’s smaller toner particulates have KSt values well over 300 bar-m/s. In some cases the protection strategies employed 10 years ago are no longer adequate. Explosion vent sizes may be too small or suppression reaction times too slow.
The decrease in particle size reduces the required energy to initiate a deflagration. It is easier to meet the conditions necessary for a deflagration or explosion by decreasing the minimum explosible concentration (MEC) decreasing the minimum ignition energy (MIE) needed to ignite the material, and decreasing the temperature at which the material may autoignite (AIT). A smaller particulate also creates a faster and stronger explosion since this greatly increases the maximum rate of pressure rise (dP/dt)max. It may also result in a more powerful pressure wave. All of which makes the material more hazardous.
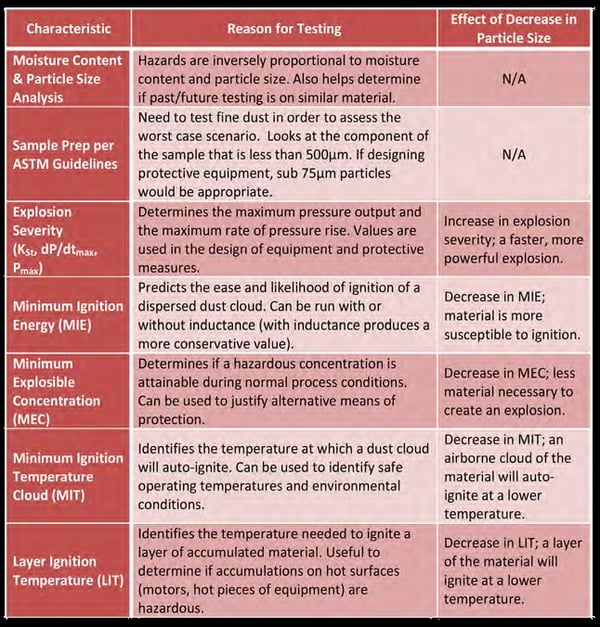
Particle size may also “unexpectedly” or inadvertently be decreased by process changes or improvements, some even as inconspicuous as replacing mill components or suppliers. For these reasons it is important to understand the impact that smaller sizes will have on your material. A summary of the recommended testing campaign and the effects of particle size on the results are provided in Table 1.
Table 1 – Recommended Tests for Material Hazard Characterization
For more information regarding hazardous or combustible dust, dust management services or other process safety concerns, please contact info@fauske.com. 630-323-8750.

